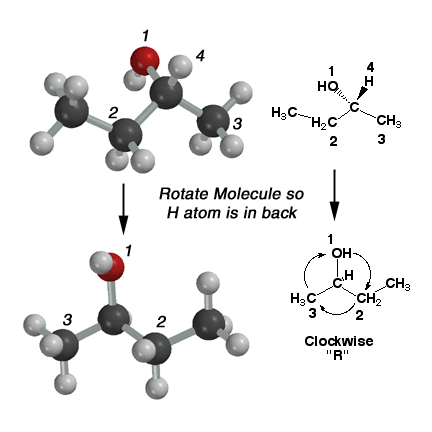
|
2-Butanol
can exist as two possible stereoisomers. More specifically, the two stereoisomers
are enantiomers of each other. Because they are stereoisomers, they are
different molecules and it is necessary to distinguish the two molecules
using the "R"
and "S" nomenclature system. In order to designate a chiral center as "R"
or "S", we must label the substituents around the chiral carbon atom in
order of increasing priority. We use the rules a) atomic weight wins, b) look
for the FIRST point of difference (this is most student's source of error)
and c) count multiple bonds as just more single bonds (look in the book for
a better description of this). In the case of 2-butanol above, the hydroxyl
group is most "important" and labeled #1, and the hydrogen atom is least "important"
and labeled #4. Next is the ethyl group (#2) followed by the methyl group (#4).
The molecule is rotated so the lowest ranking group (#4, the H atom) is pointed
away from you. Count the numbers in order #1 - #2 - #3. This motion is clockwise
in the above molecule, therefore this is R-2-butanol. If the direction had
been counterclockwise, it would have been S-2-butanol. |