|
|
|
|
|
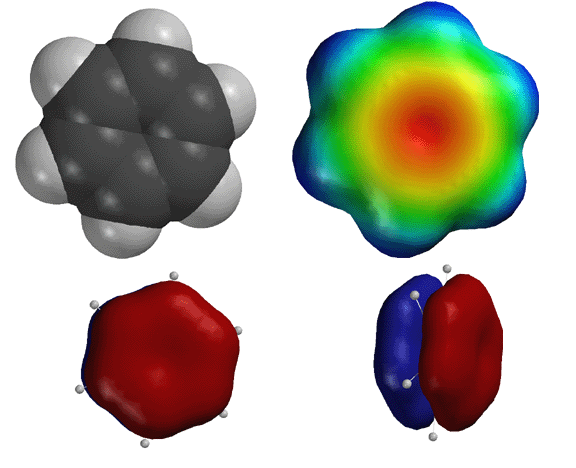
Above
are representations of benzene. The "flower" representation
on the top right is an electrostatic potential energy surface that answers
the most important question in chemistry, namely where are the electrons
in benzene. The red color indicates the region of high electron density
that is focused on the pi system. The blue color indicates the area of lowest
electron density. This picture makes sense since the pi electrons, depicted
in the lower two pictures, are exposed to solution, not surrounded by atomic
nuclei like electrons in a sigma bond. Thus, these electrons provide for
the red color, and they are able to interact with extremely strong (wicked
strong) electrophiles. Benzene should be considered a very weak nucleophile. |