|
|
|
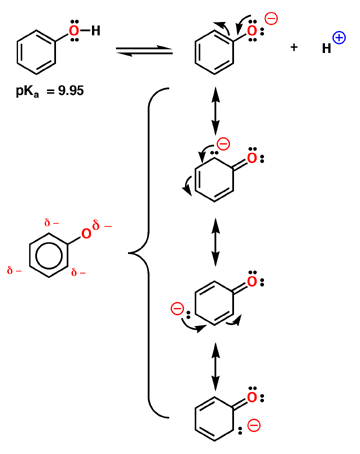
Phenol
is relatively acidic, with a pKa near 9.95. Compare this to a normal alcohol
in which the pKa is closer to 16. Phenol is more acidic because the negative
charge of the anion is delocalized around the aromatic ring as described
by the 4 contributing structures above. Note that most of the charge is still
on oxygen, but enough is delocalized in the aromatic pi system to make a
substantial difference. The interesting prediction
of these resonance contributing structures is that the negative charge is
not distributed evenly around the ring, but remains primarily in the ortho
and para positions. This is shown in the electrostatic potential surface
in which the negative charge (red color) is primarily ortho and para to the
oxygen atom. |